
Air pollution and passenger vehicles
With motorised vehicles the focus is often on reducing their carbon footprint by changing how they are powered. But what about their impact on air pollution?
Summary: Transportation, in particular road transportation is a major man-made source of air pollution. Air pollution can impact health and ultimately productivity. So how will the changing vehicle landscape impact that?
Why this is important: With motorised vehicular transportation, much of the focus has been on their global warming potential. A little closer to home, the impact of pollution, not just from the drive trains but also from other sources needs to be considered.
The big theme: The implications of air quality in general, and pollution in particular, have various significant investment and decision-making considerations. Decreasing pollution with the aim of decreasing human illness and suffering is (should be) a goal unto itself. In practice it is also important to consider how such initiatives (that more often than not increase costs and dig into profits), will be beneficial to business in the long run, whether it be through decreased healthcare costs, increased health and productivity of the workforce, and reputational gains for such decisions.

The details
Air pollution is the presence or release of a harmful substance in the air. Air is comprised mostly of nitrogen (about 78%), oxygen (about 21%) and small amounts of other gases including carbon dioxide. Pollutants are things that shouldn't normally comprise air and can be a mixture of both solid particles and gases. The most common pollutants are:
PMx = Particulate matter of varying sizes. These are tiny pieces of solids or liquids in the air. the 'x' denotes the average diameter of the particle. PM2.5 particles are 2.5 microns wide or 1/400 of a millimetre. PM2.5 is more likely to get into and deposit on the surface of the deeper parts of the lung. They can cause breathing difficulties but also a causal link to lung cancer has been found. PM10 are bigger at 10 microns wide and more like typical dust, irritating eyes, nose and throat.
NOx = Nitrous oxides typically a catch-all for nitric oxide (chemical symbol NO) which is odourless and colourless and nitrogen dioxide (NO2) which has a sharp smell and is a reddish-brown colour. NO reacts with ozone in the air to form NO2. Low levels can cause irritation, coughing, shortness of breath and nausea with consistent exposure over a few days leading to fluid build-up in the lungs. Combustion in air (e.g. burning fuels) will produce NOx.
Ozone = Ozone is a gas whose molecules are made up of three oxygen atoms (O3), rather than the normal two that we find in oxygen gas (O2). The 'Ozone layer' in the upper atmosphere protects us by absorbing most of the Sun's UV radiation. However, when ozone forms at ground level it can trigger asthma and reduce lung function. O3 can form when sunlight interacts with pollutants such as NOx, carbon monoxide and volatile organic compounds.
VOC = These are gases emitted from products and processes that can be harmful themselves or can react with other gases to form pollutants (see Ozone above). Examples include benzene, formaldehyde and toluene and sources include some solvents (e.g. paint, varnish, paint stripper), fuels, pesticides and disinfectants. As well as causing respiratory problems, they can also cause cancer.
Carbon monoxide = This is a colourless and odourless gas produced when fossil fuels are burned incompletely. The chemical symbol is CO. The biggest source in air pollution is from Internal Combustion Engine (ICE) vehicles. Once in the bloodstream, CO can make it difficult for the body's cells to bind to oxygen and that lack of oxygen causes breathing difficulties, flu-like systems and can be fatal.
SO2 = Sulphur dioxide is a colourless gas, which is very soluble in water and corrosive. It is released during volcanic eruptions but its main man-made source is from burning fossil fuels. Toxic when inhaled.
Air pollution
The link between ambient air pollution and life expectancy has been well documented. In a 2020 study conducted by the Max Planck Institute for Chemistry it was estimated that global excess mortality from all ambient air pollution was between 7.11 and 10.41 million per year. In addition it was estimated to lower life expectancy by between 2.3 and 3.5 years - more than from tobacco smoking.
A number of long term studies looking at data from Seattle and Canada have suggested a link between proximity to air pollution, particularly traffic related, and dementia risk.
There is some evidence that exam results are worse amongst students exposed to air pollution.
A proportion of PM2.5 comes from naturally occurring sources such as pollen and sea-spray. In areas such as Africa and West Asia that are close to deserts, windblown dust is a major source. Man-made sources include domestic combustion, industrial processes, manufacturing, construction and road transportation.
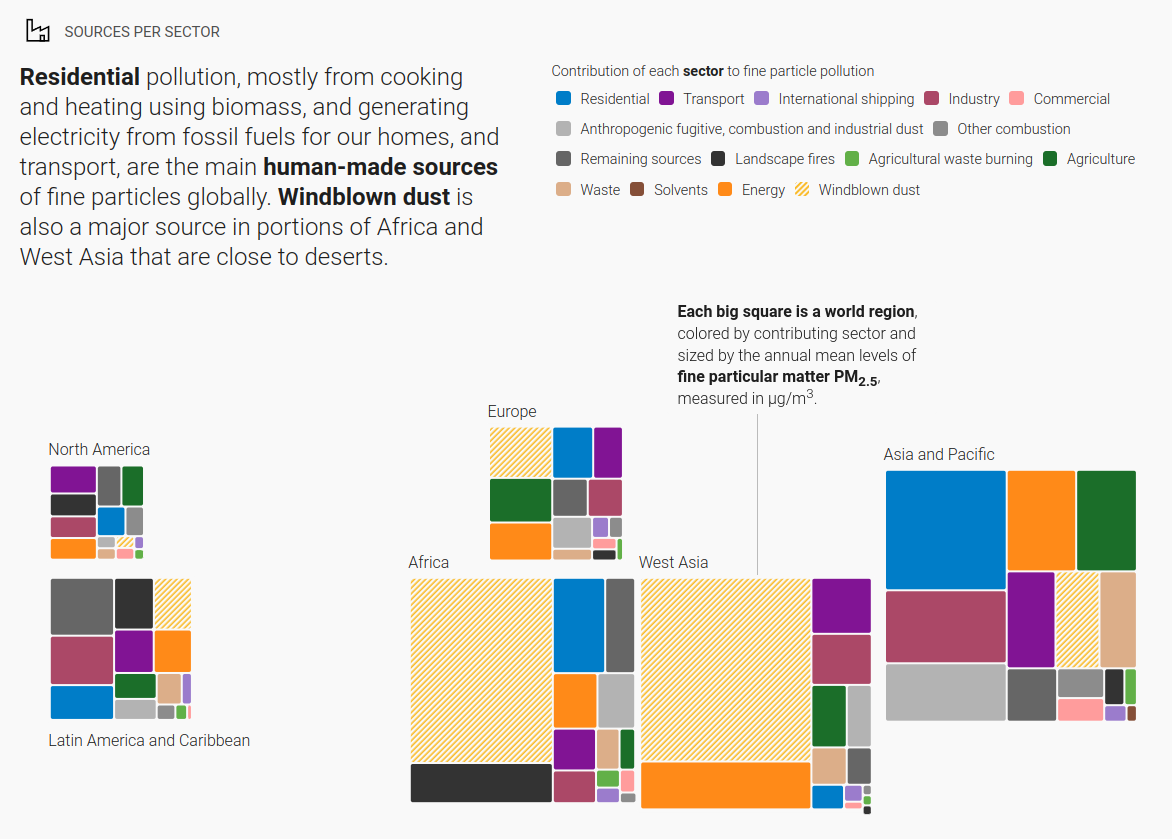
In 2021, the WHO updated their PM2.5 annual mean air quality guideline to five micrograms per cubic metre. The previous 2005 guideline level was 10 micrograms per cubic metre. This means that in 2019, 99% of the world population was living in places where the WHO’s strictest air quality guideline levels were not met.
Air pollution from passenger vehicles
There are two main ways in which passenger vehicles contribute to air pollution. The most obvious one is from their tailpipe emissions or what is created as a byproduct of converting the energy source to kinetic energy (or movement).
So far we haven't mentioned CO2 as a pollutant - strictly speaking it is not regarded as an air pollutant as it is a natural part of air. It is however a major byproduct of burning fossil fuels and has a well understood global warming potential.
EVs might be better for greenhouse gas (GHG) emissions, but a study highlights electric cars might have a less-obvious problem, in terms of air pollution, and one that is shared by most vehicles on the road.
Emissions Analytics carried out real-world emissions tests on a family hatchback and demonstrated that PM emissions emanating from tyres may actually be over 1,000 times worse than emissions from vehicle tailpipes.

While EVs do not have tailpipes, they do have tyres, and not only that, their batteries tend to make them heavier – which Emissions Analytics says could exacerbate this problem. While EV regenerative braking systems could help limit PM emissions from brake wear, the tyre issue is a problem that until now may have been dramatically underestimated.
Much of the media coverage of the Emissions Analytics report led with the headline "Pollution from tyre wear 1,000 times worse than exhaust emissions." This is however a worst-case scenario equating to 9.26 grams of PM per mile or 5.8 grams per km. A report commissioned by the RAC by Plug Life Consulting pointed out that if that were normal wear then the average driver in the UK would have bald tyres within less than 1,358 miles! Reducing tail pipe emissions are still absolutely key for both air quality and GHG emissions.
However, tyre wear is still a real issue. Emissions Analytics points out the correlation between vehicle weight and PM from tyres applies across the board – including for SUVs. This indicates that the latter’s rising popularity could not just be having a growing impact on greenhouse gas emissions – they alone are the second-largest contributor to global emissions increases since 2010 – but also a disproportionate effect on air quality.
The issue of weight in EVs relative to ICE vehicles is also perhaps a red herring. The top selling passenger car globally is the Toyota Corolla with a kerb weight (the car plus equipment fluids and fuels but not passengers or cargo) of 1,360 kg to 1,445 kg. The second top selling vehicle globally in 2022 was the Ford F-150 pick up whose lightest model has a kerb weight of 1,846 to 2,111 kg. The top selling EV globally is the Tesla Model Y weighing in at 1,909 to 1,930 kg. Much of a muchness.
Ultimately it points to a rethink about what we drive and how we drive. More on that in the 'Other things to think about' section below.
Number of vehicles
The number of passenger vehicles globally on the road is not far off one-and-a-half billion with almost 37% in Asia.
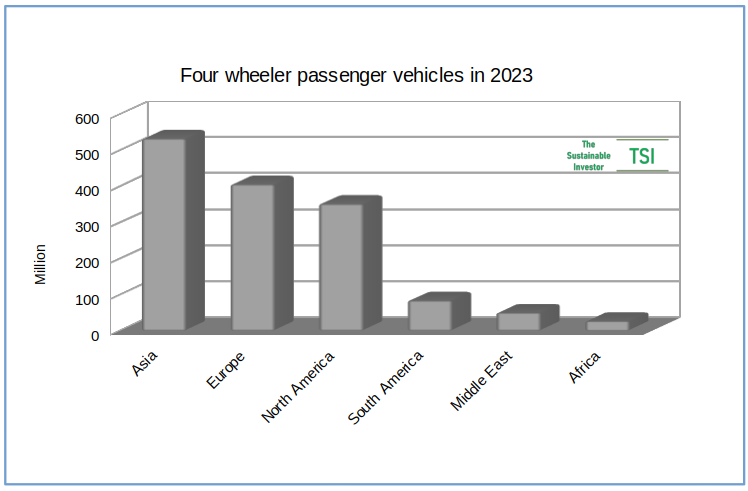
Goldman Sachs forecasts that there will be almost 120 million vehicles sold per year in total across all drive trains by 2040 up about 20% on current sales.
What about EVs?
They have been growing strongly in major markets becoming meaningful shares of total passenger vehicles sold.
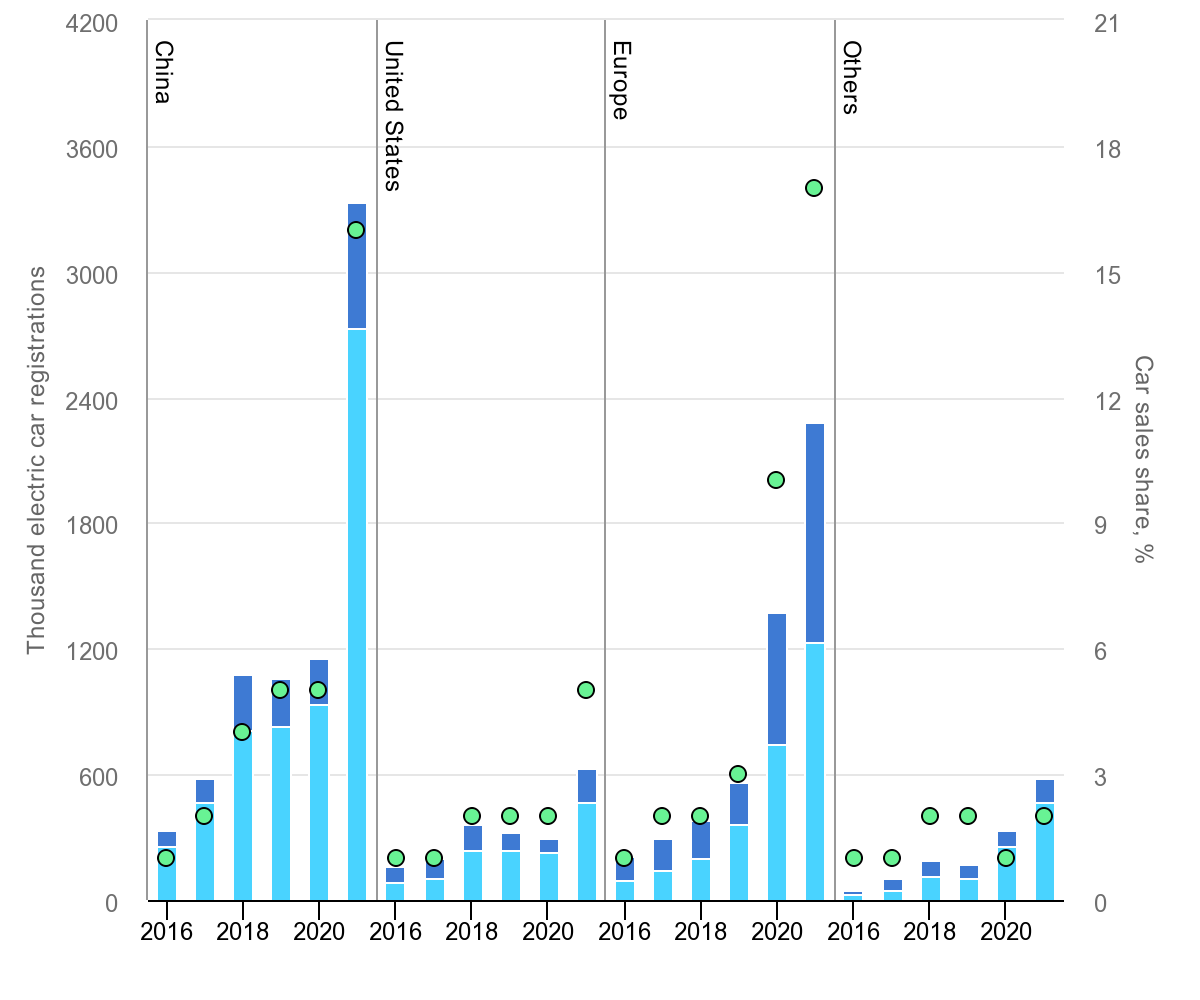
Goldman Sachs forecast that by 2035, half of new car sales worldwide will be EVs. The EU is expected to be at 100% by that point driven by legislation, with the US above 70% and China above 55%. China dominates current EV sales with around six million expected to be sold in 2023. By 2040 EV Sales globally are expected to be 73 million units per annum.
Other things to think about
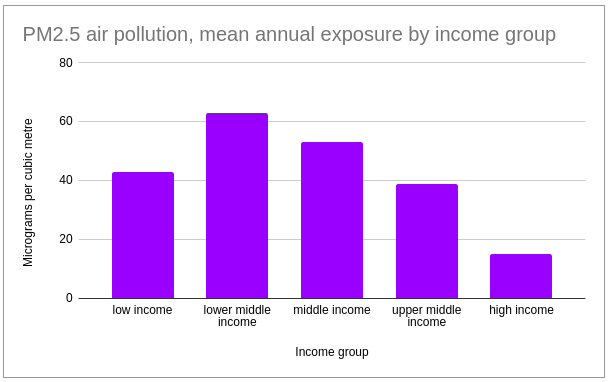
Lower middle income countries have been found to have the highest mean annual exposure to PM2.5 pollution.
Two and three wheelers are the fastest growing transport mode in many low and middle-income countries. Many of these ICE two and three wheelers are old and inefficient, producing lots of pollutants including PM. Two-stroke scooters, for example, produce more PM than an average passenger car. Moving two wheelers and three wheelers to EV makes sense as a priority in these regions therefore.
There are a lot of motorised two wheelers!
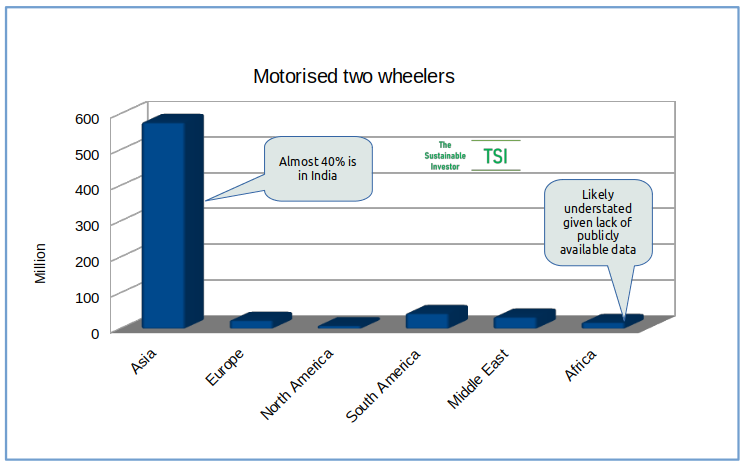
Scenario calculations using the UN Environment eMob calculator show that if we moved new motorcycle sales to 90% EV by 2030, CO2 emissions could be reduced by close to 11 billion tons between now and 2050. From a financial perspective, savings from lower fuel and maintenance costs, even taking into account a higher purchase price of EV motorcycles could add up to US$ 350 billion by 2050. And of course the positive impact on air quality.
So two/three wheelers are really important. And they could be important in developed markets as well, as we transition toward a lower car use future in our biggest cities. 👇🏾

Reducing the requirement for motorised transportation in the first place would reduce emissions too and clearly one way to do that would be to provide efficient low polluting / low emissions transport or indeed reduce the distances required to travel. This is where urban planning could become a key tool for decarbonising transportation. 👇🏾
Air pollution can be found underground too. Most of the particulate matter on the Underground is created as wheels, tracks and brakes grind against each other and very small metallic particles are thrown up. 👇🏾

Something a little more bespoke?
Get in touch if there is a particular topic you would like us to write on. Just for you.
Contact us
Please read: important legal stuff.


